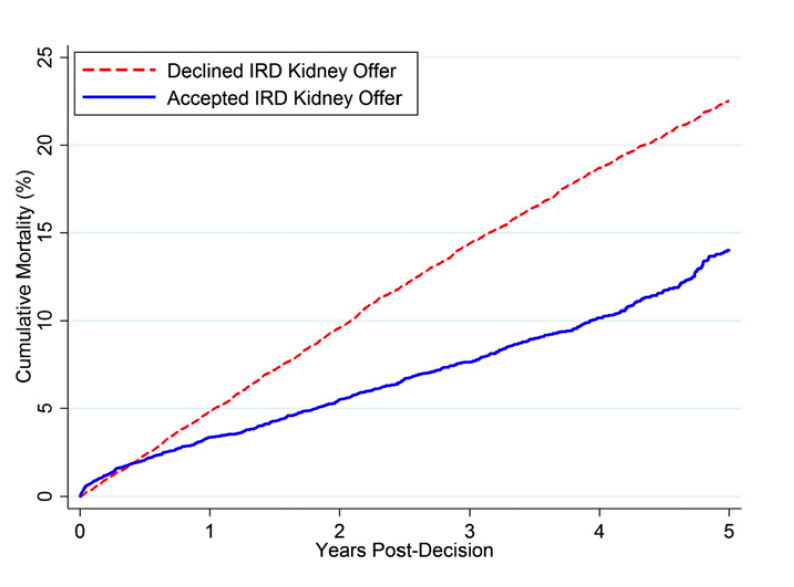
A 35 year-old Caucasian male presents with persistent swelling of both legs associated with dark colored urine for two months. He went to an emergency room 2 months ago for these complaints and was told that he has some protein and blood in the urine. He was treated with 3 days of levofloxacin. There is no other past medical history. No history of skin rash or joint swelling. On examination the blood pressure was 130/85 mm Hg and there was bilateral 1+ pedal edema. Rest of the physical examination was normal. Urine analysis showed 3+ proteinuria, 10-15 RBCs per high-power field, and occasional RBC cast. The BUN was 10 mg/dL, serum creatinine was 0.9 mg/dL. Antistreptolysin was negative, C3 level is decreased and C4 level is normal. Antinuclear antibodies, ANCA, hepatitis B and C serology were negative. 24-hour urine collection showed 2 g proteinuria and a kidney biopsy was performed. On light microscopy, kidney biopsy showed increase in the mesangial matrix and cellularity and glomerular basement membrane appeared irregularly thickened. Silver stain revealed duplication of glomerular basement membrane in multiple glomeruli. Immunofluorescence showed positive staining for C3, but negative for IgG, IgM and IgA. Electron microscopy revealed electron-dense deposits in the mesangium and sub-endothelial area.
What is the most likely diagnosis?
| A. Acute poststreptococcal glomerular nephritis | |
| B. Diffuse proliferative lupus nephritis | |
| C. Dense deposit disease | |
|
|
D. C3 glomerulopathy |
| E. |
|
|
Copyright © ABIM Exam World
Created On: 09/12/2017
Last Modified: 03/07/2021
 Omitted
Omitted